 It turns out that the strengths of these types of interactions depend upon several factors, including shape of the molecule, surface area, and number of electrons. Now you may ask yourself, why does H 2 boil at a higher temperature than He? Good question! The answer is addressed by the web activity on interactions. On the other hand, to break the van der Waals interactions between H 2 molecules, the system temperature only needs to rise to ~20K, above this temperature H 2 is a gas the interactions between individual H 2 molecules are not strong enough to resist the kinetic energy of colliding molecules. To break a H-H covalent bond, you will need to heat the system to around 5000K. We can compare energy associated with the H-H covalent bond and the H 2 – H 2 van der Waals interaction. These H-H molecules are themselves attracted to one another through LDFs, leading to van der Waals interactions. 51 This energy can take several forms: energy delivered by molecular collisions with surrounding molecules or electromagnetic energy due to the absorption of light are some of the ways covalent bonds can be broken, and we will return to them later.Įach H can form only a single covalent bond, leading to the formation of H-H molecules. 
In order to separate the hydrogen molecule back into two atoms, that is, to break the covalent bond between them, we have to supply energy. Because of its radically different stability, the H-H system gets a new name, it is known as molecular hydrogen or H 2, and the interaction between the H atoms is known as a covalent bond. This leads to a potential energy minimum for the two interacting hydrogen atoms that is much deeper than that for He-He. In an H-H interaction, the atoms are held together by the attraction of each nucleus for both electrons. As two hydrogen atoms approach one another, they form a much more stable interaction (about 1000 times stronger than the He-He van der Waals interactions. Now let us contrast the behavior of helium with that of hydrogen (H). 
The lack of stable interaction at these “higher” temperatures means that helium atoms do not stick together above 4K helium is a gas at temperatures above 4K. Meaning that at all temperatures above ~4K, there is enough kinetic energy in the atoms of the system to disrupt the van der Waals interactions between He atoms. This energy is supplied by collisions with other He atoms. In the case of He, the interaction potential energy drop is quite small (that is: the stabilization due to the interaction) and it does not take much energy to knock the two atoms apart. When two atoms of helium approach each other LDFs come into play and a van der Waals interaction develops. One advantage of focusing on molecular hydrogen and helium is that it also allows us to introduce, compare and briefly consider (we will do much more considering later on) both van der Waals interactions and covalent bonds. 
We will consider more complicated atoms and molecules after we discuss atomic structure in greater detail in the next chapter. Hydrogen atoms have 1 proton and 1 electron, Helium atoms have 2 protons and 2 neutrons in the nucleus, and 2 electrons in their electron clouds. For simplicity, we begin by considering interactions between the simplest atoms hydrogen (H), and helium (He), and the simplest molecule - molecular hydrogen (H 2). 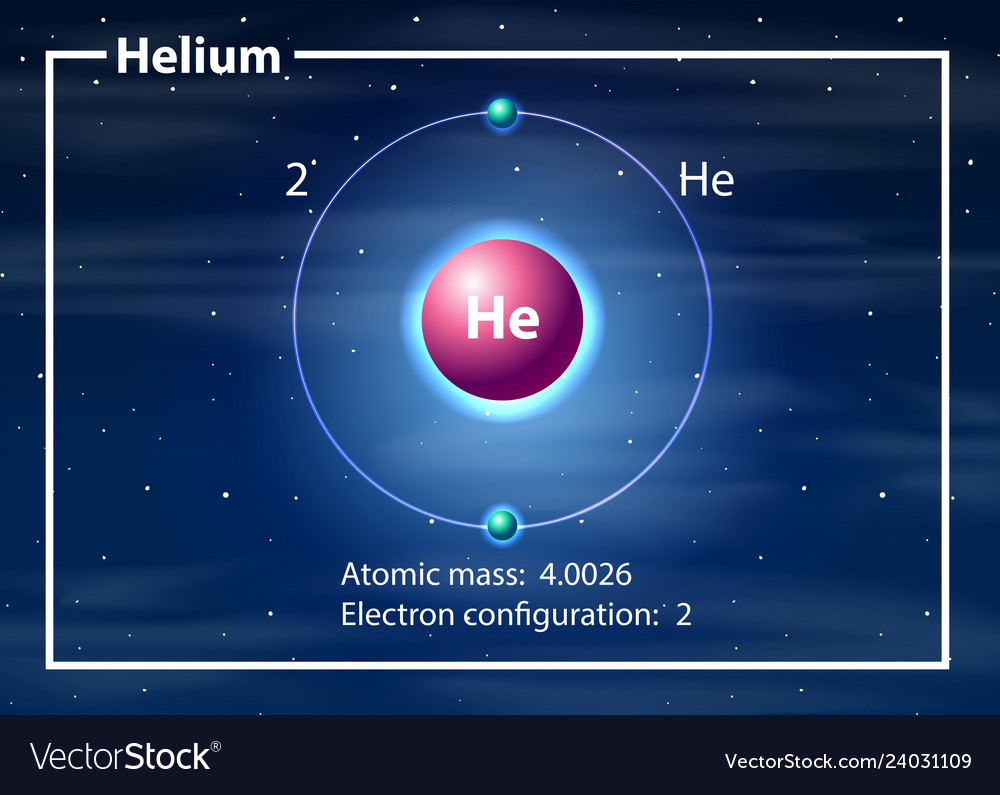
Now lets take a look at a couple of real systems. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
